 A more sophisticated treatment of bonding is needed for systems such as these. Instead, many of these species, including SrF 2 and BaF 2, are significantly bent. More disturbing, the VSEPR model predicts that the simple group 2 halides (MX 2 ), which have four valence electrons, should all have linear X–M–X geometries. 
The hybrid orbital picture, although more complex, provides a better explanation of such things In this section we will make the connection between hybrid orbital described in Chapter 6.2 and VSEPR. In fact, structural studies have shown that the H–S–H and H–P–H angles are more than 12° smaller than the corresponding bond angles in H 2 O and NH 3. It predicts, for example, that H 2 S and PH 3 should have structures similar to those of H 2 O and NH 3, respectively. 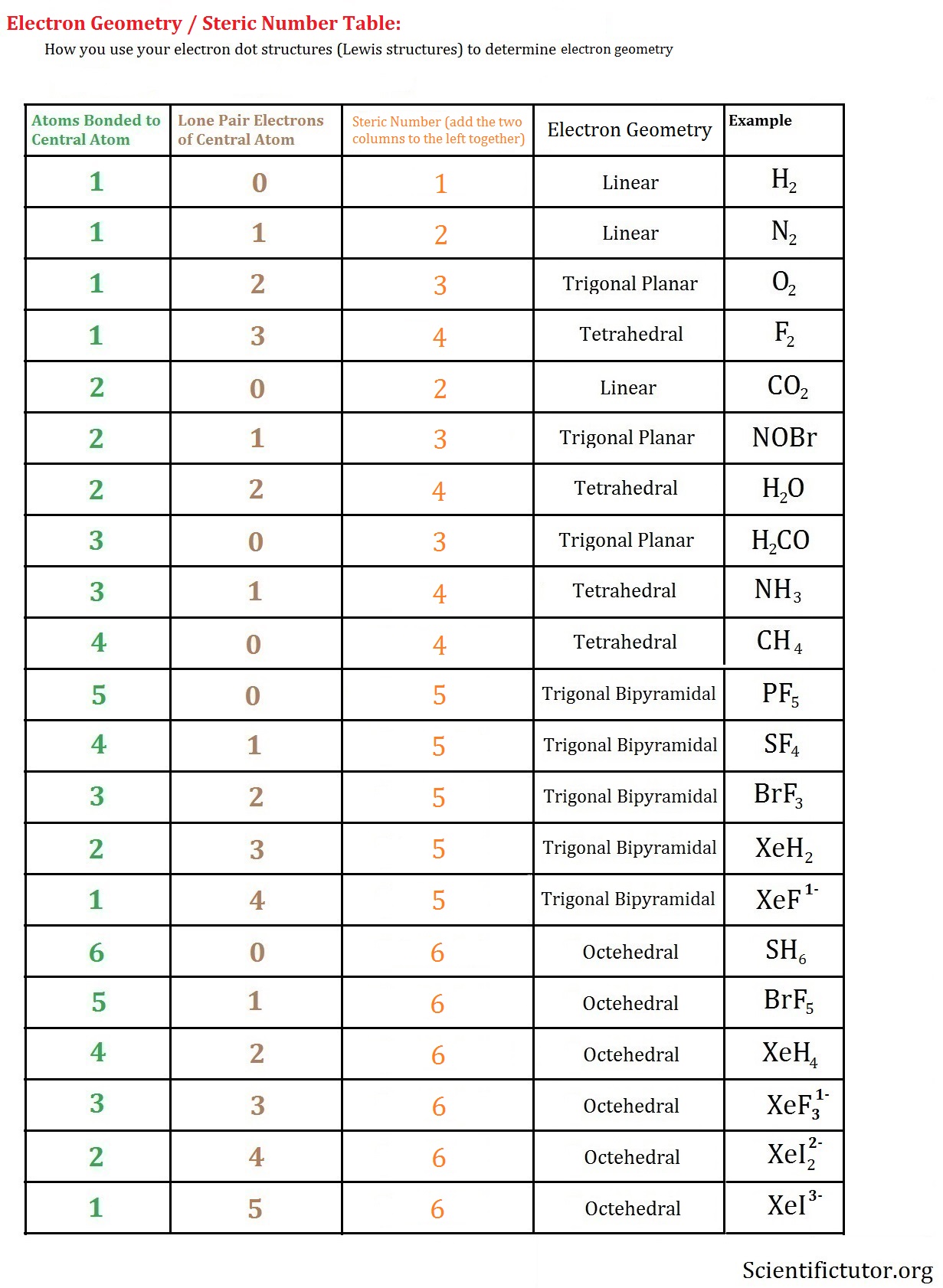
Although the VSEPR model is a simple and useful method for qualitatively predicting the structures of a wide range of compounds, it is not infallible. Keep in mind, however, that the VSEPR model, like any model, is a limited representation of reality the model provides no information about bond lengths or the presence of multiple bonds. (pronounced “vesper”), which is simple to use to predict the shapes of many molecules and polyatomic ions. We continue our discussion of structure and bonding by introducing the valence-shell electron-pair repulsion (VSEPR) model A model used to predict the shapes of many molecules and polyatomic ions, based on the idea that the lowest-energy arrangement for a compound is the one in which its electron pairs (bonding and nonbonding) are as far apart as possible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
